Applying the Trends of Oxidation States in the Periodic Table
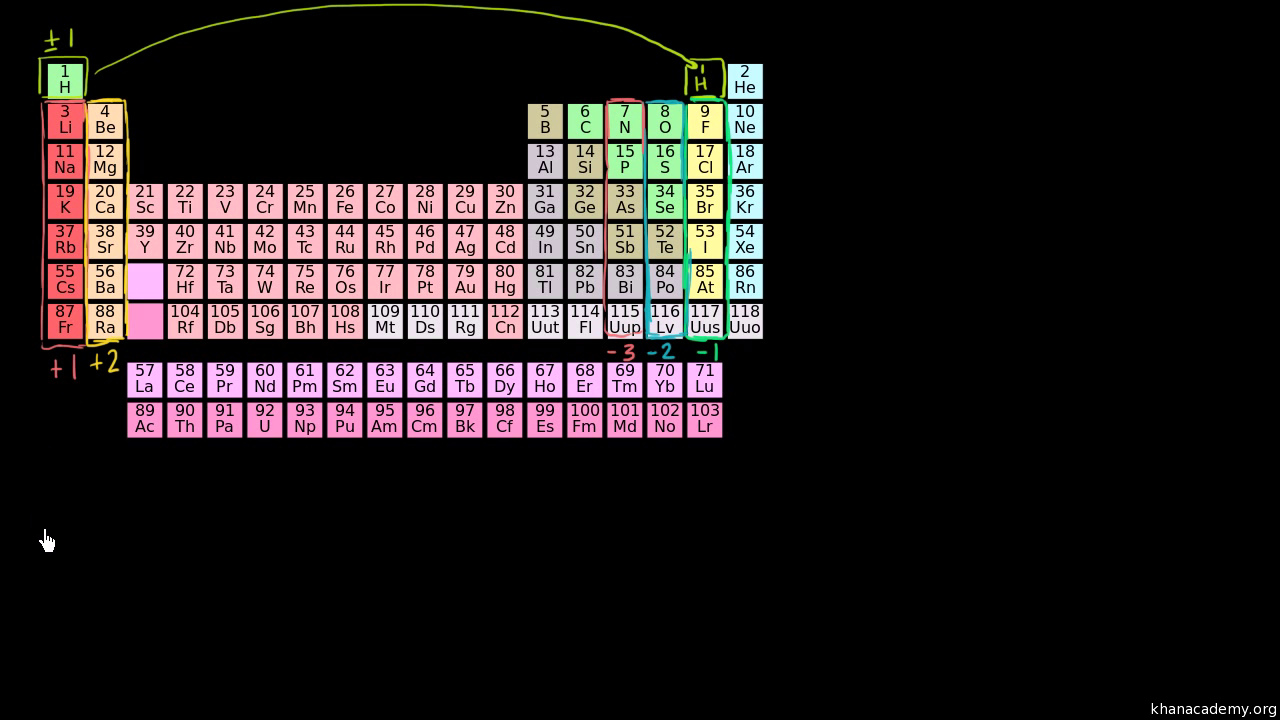
Well Im going to box-- Im going to separate hydrogen because its kind of a special case. So first lets just focus on the alkali metals.

Solved Applying The Trends Of Oxidation States In The Chegg Com
B transition metals and non metals.

. 2 N Na Mg SCORE. Remember you can revisit the oxidation number image in the Media Tab of the Lab Pad a -1 b 7 c -7 d 1. Alkaline earths and coinage Cu Ag Au metals.
Know group and period general trends for atomic radius. Remember you can revisit the oxidation number image in the Media Tab of the LabPad a -1 b 7 c -7 d 1. Most metals tend to easily lose their valence or outermost electrons giving them positive oxidation states while non-metals in general tend to accept electrons better resulting to negative oxidation states.
Larger elements in this group exhibit multiple oxidation states. Ionic compounds show oxidation accordi. Consider a carbon atom for example.
Applying the trends of oxidation states in the periodic table what is a likely oxidation state of Chlorine. 72160 DAY1 0812 AM PROGRESS. A simple rule to remember is the oxidation state is for unmixed atoms the same as their charge.
Apply trends to arrange elements in order of increasing or. Periodic Table of the Elements With Oxidation Numbers. Sulfur is another p block element which has different oxidation numbers-2.
Applying Knowledge Of Empirical Formulas Iron Oxides And. But if we look at the alkali metals the Group 1 elements right over here weve. Metalloids and alkali metals.
Values in italics represent theoretical or unconfirmed oxidation numbers. All of the non-metals in the table on the other hand have at least one common negative oxidation state. The Periodic Table of Oxidation States.
This periodic table contains the oxidation numbers of the elements. The Group 3A13 elements can be divided into two categories based on their physical and chemical properties. Common oxidation states are in bold.
The elements of group IA to IVA have the same oxidation states as their group numbers are. Well think about hydrogen in a second. H 2 SO 4 BaSO 4.
Jul 09 2021 0254 AM. HOME THEORY MEDIA MISSION Applying the trends of oxidation states in the periodic table what is a likely oxidation state of Chlorine. This table is based on Greenwoods1 with all additions noted.
This periodic table in PDF format can. The arms in a Spiral periodic table are for Choose all that apply. Most common oxidation number of oxygen is -2.
Transition metal often have a wider range of possible oxidaions states. The only consistent variation of oxidation numbers with the Periodic Table is that in their compounds Group 1 metals are always 1 and Group 2 metals are always 2. Example for -1 oxidation state is hydrogen peroxide H 2 O 2-2.
A simple rule to remember is the oxidation state is for unmixed atoms the same as their charge. Alkali metals and noble gases. This last one shows the various known oxidation states for the different elements in the periodic table.
Important trends generally followed in the periodic table are electronegativity ionization energy electron affinity atomic radii ionic radii metallic character and chemical reactivity. Webelements periodic table iron oxide how to write the formula for iron iii sulfate you periodic table with common ionic charges what is the molar mass of iron iii oxide fe2o3 quora. Oxidation State Trends In Periodic Table Khan Academy.
Just as B Al and Ga come from group IIIA for this reason they constantly reveal an oxidation state of 3. Still if you are curious as to what it depicts theres more detail in the original post. SO 2 H 2 SO 3 6.
Bold numbers represent the more common oxidation states. It is basically the charge on an atom in a compound. This fantastic video tells you that how the group number in the peri.
Because of the way that we count electrons for oxidation number purposes carbon can have any of the oxidation numbers. Oxidation states One way of taking into account the number of electrons being lost or gained in a redox reaction is by looking at an elements oxidation state or its oxidation number. 100794 6941 2-1 Periodic Table of the Elements Group 18998403 F 2-7 35453 17 2-8-7 79904 35 2-8-18-7 126905 53 2-8-18-18-7 210 85.
This video tells that how oxidation State very and what is its trend in periodic table. The common oxidation states of all of the metals in the periodic table are all positive. This table also contains the element number element symbol element name and atomic weights of each element.
Its probably of more use to the chemists out there than for general interest. Up to 24 cash back Chm132 Infer the physical properties atomic radius metallic and nonmetallic characteristics of an element based on its position on the Periodic Table. Main group elements have usually less numbers of oxidations states.
For example chromium can have an oxidation state of either 3 or 6 while manganese can take 2 3 4 6 and 7 as its oxidation states. Or some general trends for oxidation states by looking at the periodic table. Ä1 H 1 He Li 1 Be 2 Ä1 B 1 2 3 2 Ä4 Ä3 Ä2 Ä1 C 1 2 3.
Selected Oxidation States Relative atomic masses are based on 12C 12000. A simple rule to remember is the oxidation state is for unmixed atoms the same as their charge. The format of the table based on one devised by Mendeleev in 1889 highlights some of the periodic trends.
Oxidation state 0 which is found for all elements is implied by the column with the elements symbol. 120 rows Oxidation State of all the elements in the Periodic Table in Graph and Table format. The oxidation state of a normal element is directly or indirectly related to the group number to which the element belongs in the table of elements.
The oxidation state of elements are largely dependend on their position in the periodic table. The Hull Periodic Table points out correlations between. Applying the trends of oxidation states in the periodic table what is a likely oxidation state of Chlorine.
The d block metals shown on the table in yellow have the widest range of oxidation states. Na 2 O MgO H 2 O are examples to -2 oxidation state of oxygen. Choose the true statement.
Using the Periodic Table Define atomic radius and ionic radius. 64 33 Interact with the LabPad to answer the quiz. This video is about the oxidation states of atoms in a compound.
Electronegativity ionization energy and electron affinity increase as we move from the left to right direction in the periodic table and these properties decrease as we move top to bottom. Na 2 S H 2 S. Other transition metals capable of having multiple oxidation states are labeled in the.
And Ill box them off. For example the 3 oxidation state is common for B and Al while Ga In and Tl more frequently exhibit the 1 oxidation state.
Oxidation State Trends In Periodic Table Video Khan Academy

Solved Applying The Trends Of Oxidation States In The Periodic Table What Is A Likely Oxidation State Of Chlorine A Simple Rule To Remember Is The Oxidation State Is For Unmixed Atoms The

Comments
Post a Comment